Issazadeh-Navikas Group
Neuroinflammation unit

Our team focuses on neuronal immunity, the interaction between the immune and central nervous systems—specifically, the cross-regulation between immune genes and neurons in the CNS. Ultimately, our aim is to understand the mechanisms by which these cells communicate with and govern each other. We hope to identify shared molecules that are central to the stability, integrity, and function of neurons in the CNS, which also play a central role in reprogramming pathogenic T cells that enter the CNS. These neuroimmunoregulatory genes could then be targeted therapeutically to treat and prevent neuroinflammation and neurodegenerative diseases.
IFN‐β rescues neurodegeneration by regulating mitochondrial fission via STAT5, PGAM5, and Drp1:
All cells require energy provided by mitochondria for their normal function and survival. Energy demanding neurons are particularly dependent on proper mitochondrial function, to allow their fast and rapid signaling along axons extending far from the cell soma. In a recent paper1, we report that endogenous type I interferon (IFN) signaling is essential for maintaining a healthy mitochondrial status in neurons, by allowing the separation and 2removal of damaged mitochondria via a process called mitochondrial fission. This is particularly important to also generate correctly sized mitochondria which can be transported along the axons, and therefore provide energy locally, specifically in myelinated axons. Lack of IFNb causes impaired STAT5, PGAM5 and Drp1 signaling and unsuccessful endoplasmic reticulum (ER) wrapping around the damaged mitochondria to complete fission. This leads to accumulation of huge and damaged mitochondria, which clot the axons and thereby prevents their proper energy supply and gradual degeneration. These findings have relevance for neurodegenerative diseases including Multiple sclerosis and Parkinson’s disease (IFN‐β rescues neurodegeneration by regulating mitochondrial fission via STAT5, PGAM5, and Drp1. Emilie Tresse, Lluís Riera‐Ponsati, Elham Jaberi, Wei Qi Guinevere Sew, Karsten Ruscher, Shohreh Issazadeh‐Navikas. EMBO J (2021)e106868.)
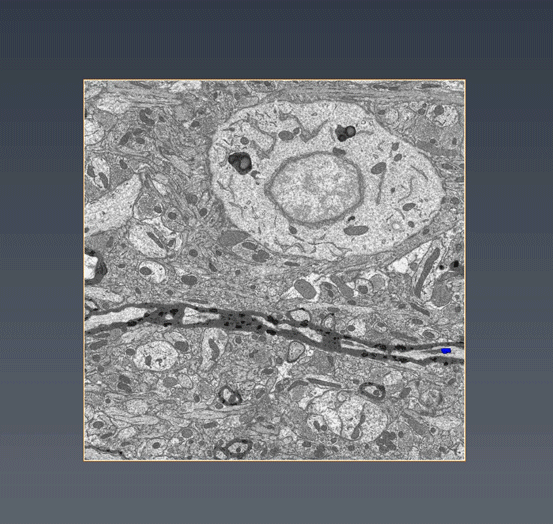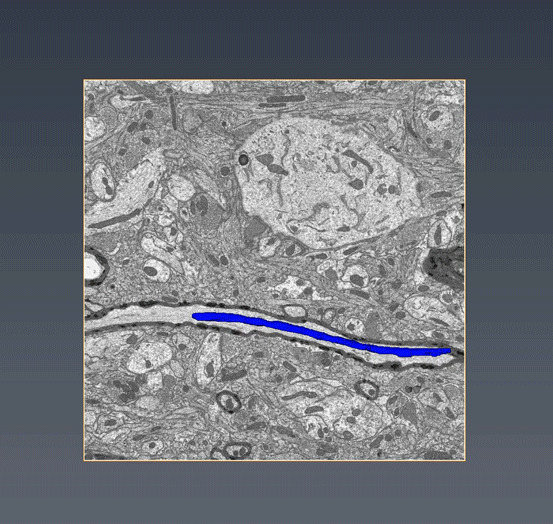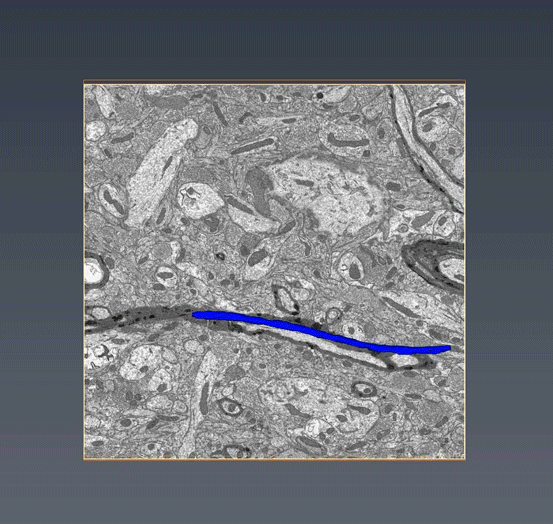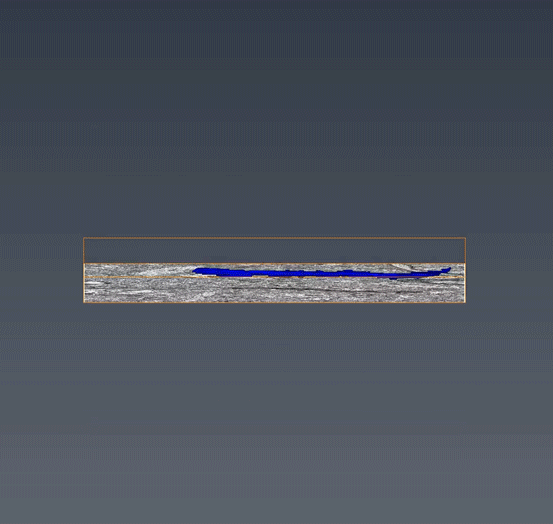
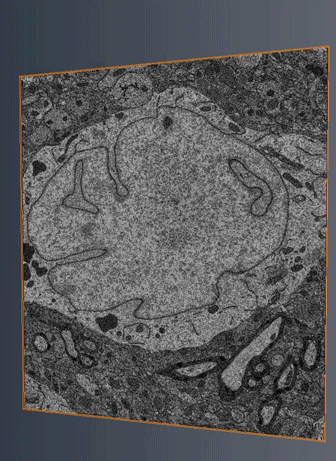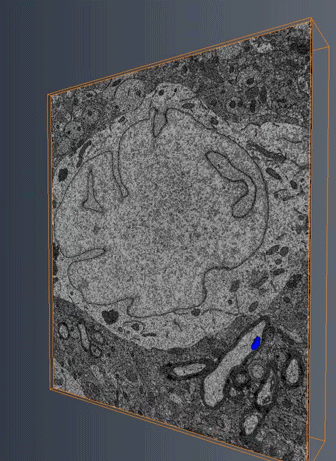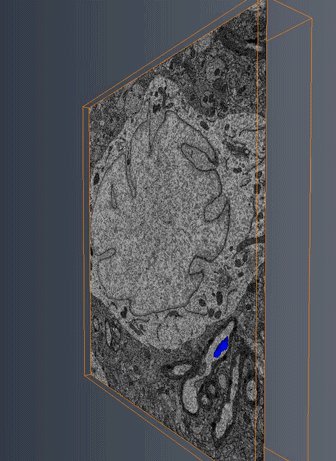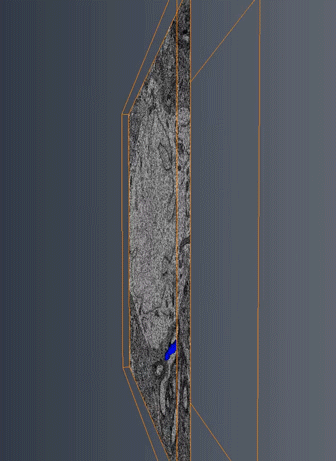
MitoSV-seq: An advance in mitochondrial DNA sequencing: We have developed MitoSV-seq, a high-resolution technique that sequences all types of variations in mitochondrial DNA in single cells, including neurons, with which we identified a hotspot of mutations that is common to neurodegeneration and several cancers (Jaberi et al. EBioMedicine 2020).
Neuronal immune genes are central in preventing neurodegeneration: We have identified that several immune genes are essential for neuronal homeostasis and function. Accordingly, we determined that the lack of the immunoregulatory cytokine IFNβ or its receptor, IFNAR1, is sufficient to cause spontaneous neurodegeneration and deficits in motor coordination and cognition in mice—resembling Parkinson disease dementia and dementia with Lewy bodies (Figure) (Ejlerskov et al. Cell 2015).
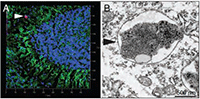
Figure. a-synuclein, a PD dementia-causing protein aggregate, accumulates in nerve cells when the Ifnb gene is lacking. A. Light microscopy image of a Lewy body (red) inside the brain of an Ifnb-deficient mouse. B. A similar protein aggregate, shown by electron microscopy (arrowhead). Based on Ejlerskov et al. Cell 2015.
Neurons tame pathogenic T cells in CNS: We have discovered that neurons are central in regulating neuroinflammation—taming pathogenic T cells and reprogramming them to become FoxP3+ T (Liu et al. Nature Medicine 2006) and FoxA1+T regulatory T cells (Liu et al. Nature Medicine 2014). The latter, FoxA1+ Tregs, are a novel population of regulatory T cells that we identified as being induced by IFNβ to prevent neuroinflammation in multiple sclerosis and its animal model, EAE.
Neuronal immune genes are essential for reprogramming encephalitogenic T cells: We have determined that neuronal expression of immunoregulatory genes, such as TGFβ and B7.1, is critical in the crosstalk between neurons and pathogenic T cells and for neuronal capacity to reprogram encephalitogenic T cells into FoxP3+ Tregs (Liu et al. Nature Medicine 2006). We have also found neuronal IFNβ and PD-L1 to be central for the generation of FoxA1+Tregs (Liu et al. Nature Medicine 2014, Liu et al. Nature Communications 2017).
Neuroinflammation is a core element in many inflammatory and neurodegenerative conditions and the leading cause of neurological diseases, including multiple sclerosis, Alzheimer disease, and Parkinson disease. By determining the mechanisms by which immunoregulatory factors control the function of neurons and neuroinflammation, we can isolate and prevent neuroinflammatory conditions from arising and thus limit neurodegeneration.
Our lab pursues research in several overarching themes in neuroimmunity using molecular, cellular, organismal, and bioinformatic approaches, from single-cell transcriptomics to murine disease models. Through collaborative efforts, many of our findings are translated to clinical populations to examine disease mechanisms and trajectories.
Regulation of inflammation in the CNS
One of our aims is to understand the immune factors that regulate inflammation in the CNS. Specifically, we are examining the function of transcription factors and signaling molecules in the plasticity and activities of immune cells, particularly FoxP3+ and FoxA1+ regulatory T cells, which we have shown suppress certain neuropathologies. These projects are performed in the context of neuronal function and homeostasis and neuroinflammatory diseases, including multiple sclerosis and Parkinson disease.
Our current focus is to establish the genetic, epigenetic, and proteomic profiles of FoxA1+ Tregs and examine the plasticity of other subsets of T cells in assuming the main genetic and functional properties of FoxA1+ Tregs by large-scale RNA-seq, scRNA-seq, ChIP-seq, and proteomics.
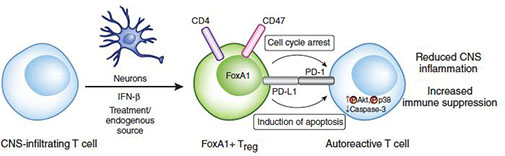
Figure 1. Neurons, via expression of IFNβ, reprogram pathogenic T lymphocytes in the brain to become anti-inflammatory cells, called FoxA1+ regulatory T cells, in MS (Liu et al., Nature Medicine 2014). Adapted from News & Views by Delgoffe and Vingali (Nature Medicine, 2014).
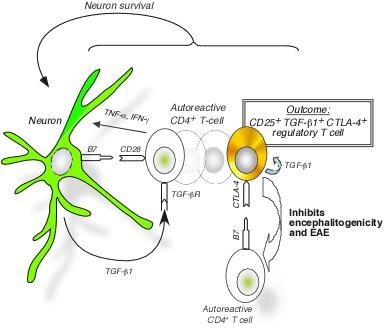
Figure 2. Neurons, via expression of TGFβ and B7, convert pathogenic T lymphocytes in the brain into anti-inflammatory cells, called FoxP3+ regulatory T cells, which prevent MS-like disease (EAE) (Liu et al., Nature Medicine 2006). Highlighted in News & Views by Fujinami (Nature Medicine 2006).
Neurodegeneration
Many neurodegenerative diseases and events are regulated by immune genes, particularly in neurons, such as the aggregation of a-synuclein. Thus, we are interested in identifying these genes and determining how they influence neuropathologies in Parkinson disease and other dementias. Our work concerns how immune genes, such as IFNβ and its downstream signaling components, control neuronal homeostasis through the regulation of autophagy/mitophagy and mitochondrial dynamics and function.
Currently, we are determining the functions of other candidate immune genes in neurons and their impact in preventing neuroinflammation and neurodegeneration. We have identified and characterized the functions of associated genes in sporadic and familial Parkinsonian disorders. Moreover, we are establishing how neurons use immune signaling to interact with other CNS-resident cells to prevent gliosis.
Selected publications
IFN‐β rescues neurodegeneration by regulating mitochondrial fission via STAT5, PGAM5, and Drp1. Emilie Tresse, Lluís Riera‐Ponsati, Elham Jaberi, Wei Qi Guinevere Sew, Karsten Ruscher, Shohreh Issazadeh‐Navikas. EMBO J (2021)e106868.
Identification of unique and shared mitochondrial DNA mutations in neurodegeneration and cancer by single-cell mitochondrial DNA structural variation sequencing (MitoSV-seq). Jaberi E, Tresse E, Grønbæk K, Weischenfeldt J, Issazadeh-Navikas S. EBioMedicine. 2020 Jul;57:102868. PMID: 32629384
ADAM12 is a costimulatory molecule that determines Th1 cell fate and mediates tissue inflammation. Liu Y, Bockermann R, Hadi M, Safari I, Carrion B, Kveiborg M, Issazadeh-Navikas S. Cell Mol Immunol. 2020 Jun 22. PMID: 32572163
Neuronal IFN-beta-induced PI3K/Akt-FoxA1 signalling is essential for generation of FoxA1+Treg cells. Liu Y, Marin A, Ejlerskov P, Rasmussen LM, Prinz M, Issazadeh-Navikas S. Nat Commun. 2017 Apr 24;8:14709. PMID: 28436428
Lack of Neuronal IFN-β-IFNAR Causes Lewy Body- and Parkinson's Disease-like Dementia. Ejlerskov P, Hultberg JG, Wang J, Carlsson R, Ambjørn M, Kuss M, Liu Y, Porcu G, Kolkova K, Friis Rundsten C, Ruscher K, Pakkenberg B, Goldmann T, Loreth D, Prinz M, Rubinsztein DC, Issazadeh-Navikas S. Cell. 2015 Oct 8;163(2):324-39. PMID: 26451483
FoxA1 directs the lineage and immunosuppressive properties of a novel regulatory T cell population in EAE and MS. Liu Y, Carlsson R, Comabella M, Wang J, Kosicki M, Carrion B, Hasan M, Wu X, Montalban X, Dziegiel MH, Sellebjerg F, Sørensen PS, Helin K, Issazadeh-Navikas S. Nat Med. 2014 Mar;20(3):272-82. PMID: 24531377
Neuron-mediated generation of regulatory T cells from encephalitogenic T cells suppresses EAE. Liu Y, Teige I, Birnir B, Issazadeh-Navikas S. Nat Med. 2006 May;12(5):518-25.PMID: 16633347
Role of passive T-cell death in chronic experimental autoimmune encephalomyelitis. Issazadeh S, Abdallah K, Chitnis T, Chandraker A, Wells AD, Turka LA, Sayegh MH, Khoury SJ. J Clin Invest. 2000 Apr;105(8):1109-16. PMID: 10772655
Researchers find a cause of Parkinson’s disease
Also published in Danish media DR, Berlingske, Politiken, etc. (in Danish): Nyt dansk studie: Parkinson spreder sig som en steppebrand gennem hjernen
5 October 2023
Cover image, EBioMedicine- July 2020 issue.
Identification of unique and shared mitochondrial DNA mutations in neurodegeneration and cancer by single-cell mitochondrial DNA structural variation sequencing (MitoSV-seq) by Jaberi et al.
New method reveals common biomarker for cancer and neurodegeneration disease
3 July 2020
Reducing ADAM12 protein can limit activation of overly active immune cells in MS
30 June 2020
Communication patterns may be key to multiple sclerosis treatment
24 April 2017
2017 interview with PhD student Louise Munk Rasmussen on TV2 News on the implications of the lab’s research for multiple sclerosis.
2017 interview with PhD student Louise Munk Rasmussen on TV2 News on the implications of the lab’s research for multiple sclerosis.

Immune gene prevents Parkinson's disease and dementia
9 October 2015
New blood cells fight brain inflammation
16 February 2014
Regulatory T cells: Mind control
Nature Reviews Immunology 2014
Glioblastoma prognosis linked to neuronal PD-L1 expression in tumour-adjacent tissue
Nature Reviews Neurology 2013
Vaccine boosts your immune system
14 December 2010
Science4ChildrenDK- An outreach initiative in which children aged 8-11 years discover science with real-life scientists through on-site visits to schools by members of the Issazadeh-Navikas lab and trips to BRIC to design and perform basic experiments and learn about microscopy.

DHL Run in Fælledparken with members of the Issazadeh-Navikas lab.

Members of the Issazadeh-Navikas lab at the 2019 BRIC Christmas party.










