A novel blood stem cell marker for improved cancer therapy
High-dose chemotherapy represents a standard therapeutic modality for more than 40.000 cancer patients world-wide with otherwise incurable, therapy-resistant cancer. However, this treatment modality is limited by toxic effects on blood-forming stem cells in the bone marrow resulting in ablation of the blood and immune system. In order to re-establish the damaged blood system after high-dose chemotherapy it is required to transplant the patients own blood stem cells to re-establish their blood system. For this, life-saving blood stem cells are first mobilized into the blood stream, harvested from the patient, frozen, and ultimately given back as soon as the cancer patients have been treated with high-dose chemotherapy and the toxic drugs have been cleared from the body.
Researchers from the Theilgaard-Mönch group at the Finsen Laboratory, Rigshospitalet and BRIC, have now identified a novel cell surface marker specific for blood stem cells (Figure 1) that can potentially improve quality assessment of blood stem cell content in transplantation products collected from cancer patients. Given that unmanipulated transplantation products contain varying numbers of contaminating cancer cells that can cause relapse of their disease, the researchers propose that their novel blood stem cell marker can be applied for the purification of blood stem cells from the unmanipulated transplantation products allowing for production of cancer cell-free transplantation products that will reduce relapse rate and ultimately improve survival of cancer patient. These promising results have just been published in Blood Advances.
Finding the one marker that alone allows for a better purification of blood stem cells is really exciting, and important in the clinical setting, where simple and fast protocols are needed for improvement of quality assessment and manufacturing of blood stem cell products. Endomucin stands out as a more specific marker for HSCs than the currently widely used marker CD34, and even more remarkable is that Endomucin seems to be absent on cancer cells, which is a prerequisite for designing novel protocols aiming separating blood stem cells from cancer cells in order to manufacture cancer cell-free transplantation products. Kristian Reckzeh, Assistant professor
Theilgaard Group, Finsen Lab
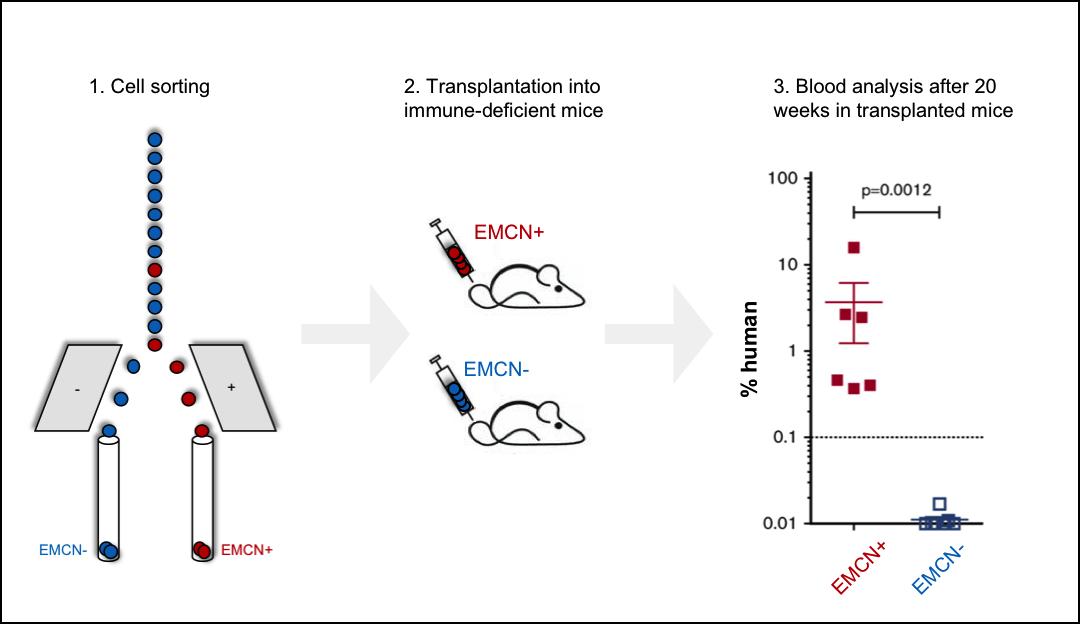
Figure 1. Endomucin is new marker of human blood stem cells. (1) Using dye-conjugated antibodies against cell surface markers, human bone marrow cells were sorted into red-stained Endomucin expressing cells (EMCN+) and unstained cells lacking expression of Endomucin (EMCN-). (2) 2000 EMCN + and 50000 EMCN- cells were transplanted into a cohort of mice. (3) After 20 weeks, analyses of bone marrow samples demonstrated the presence of mature human blood cells in mice transplanted with EMNC+ but not EMCN-, implicating that all long-term repopulating blood stem cells with the ability to generate mature human blood cells after 20 weeks are EMCN+.
Contact
Kristian Reckzeh
Assistant professor
Theilgaard Group
Email: kristian.reckzeh@bric.ku.dk
Phone: +45 35 45 60 56
Anne Rahbek-Damm
Communications Officer
Biotech Research & Innovation Centre
Email: anne.rahbek@bric.ku.dk
Phone: + 45 21288541
Publication
Human adult HSCs can be discriminated from lineage-committed HPCs by the expression of endomucin,
Kristian Reckzeh, Hüsün Kizilkaya, Alexandra Søgaard Helbo, Montserrat Estruch Alrich, André Gundersen Deslauriers, Amit Grover, Nicolas Rapin, Fazila Asmar, Kirsten Grønbæk, Bo Porse, Niels Borregaard, Dietmar Vestweber, Claus Nerlov and Kim Theilgaard-Mönch
Blood Advances 2018 2:1628-1632; doi: https://doi.org/10.1182/bloodadvances.2018015743
