Erler Group
Decoding the Matrix to Defeat Metastasis.

Metastasis remains the ultimate barrier to curing cancer.
We focus on the extracellular matrix (ECM) and the tumour microenvironment, which together create fertile soil that enables cancer cells to colonise and grow at distant organs.
By uniting fundamental biology, advanced imaging, and patient‑derived models with clinical workflows, we operate at the cutting edge of precision medicine and translational innovation. Ultimately, we aim to reveal the mechanisms driving metastatic colonisation and convert these insights into life-saving therapeutic strategies.
- I. Tumour Microenvironment & ECM Biomechanics
- II. Precision Cancer Medicine
- III. Therapeutic Innovation & Drug Discovery
I. Tumour Microenvironment & ECM Biomechanics
We dissect the biochemical and mechanical cues within the ECM that drive tumour progression, metastatic colonization, and drug resistance. By understanding how the ECM is altered in disease, we can identify how cancer cells respond to these changes to become more aggressive.
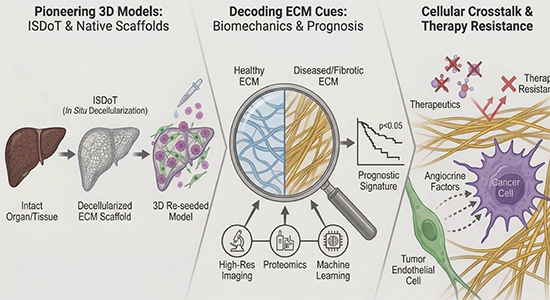
Pioneering 3D Models
Our lab developed ISDoT (In Situ Decellularization of Tissues), a groundbreaking method to isolate intact 3D ECM scaffolds from both animal organs and human tissue (Mayorca et al, 2017, Nature Medicine; Mayorca et al, 2019, Nature Protocols).
This technology allows us to accurately model metastatic environments , culture cells within native scaffolds, and assess their behavior in healthy versus diseased conditions (Rafaeva et al, 2022, Advanced Healthcare Materials).
Decoding ECM Cues
By coupling high-resolution imaging, proteomics, and machine learning, we uncover hidden features of disease. We have shown how tissue fibrosis, basal membrane stiffness and integrity actively promote cancer cell proliferation, metastasis and drug response (Cox et al, 2013, Cancer Research; Reuten et al, 2021, Nature Materials; Nielsen et al, 2021, Nature Communications; Xia et al, 2025, Frontiers in Immunology), and we have derived a desmoplastic signature capable of predicting patient prognosis. (Emerson et al, 2025, Acta Biomaterialia).
Cellular Crosstalk & Therapy Resistance
We investigate the complex dialogue between cancer cells and their surroundings. This includes identifying how specific ECM components significantly reduce a tumour's response to drugs, and discovering how Tumor Endothelial Cells (TECs) actively drive cancer aggressiveness by secreting specific angiocrine factors (Oria et al, 2025, Cell Commun Signaling; Righetti et al, 2025, IJMS).
II. Precision Cancer Medicine
In close partnership with clinical teams, we integrate real‑time molecular profiling with patient‑derived organoids and advanced co‑culture platforms to predict treatment response. We also define postoperative therapeutic windows where targeted interventions and lifestyle changes can alter disease trajectory.
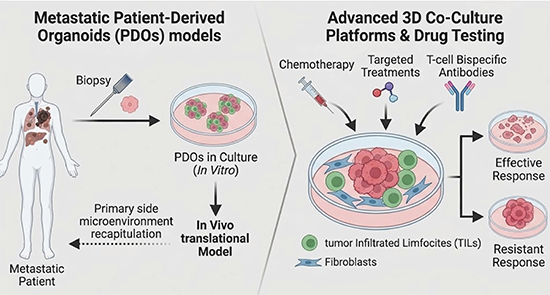
Metastatic Patient-Derived Organoids (PDOs)
We use PDOs to create personalized, laboratory models of a patient's metastatic cancer. We have demonstrated that while PDOs faithfully replicate the features of the original tumour, engrafting these PDOs into in vivo models successfully restores the complex, context-dependent microenvironmental signalling lost in isolation, providing us with a highly accurate platform for functional precision medicine (Castro et al, 2026, Frontiers in Bioengineering and Biotechnology).
Advanced 3D Co-Culture Platforms
We have developed a state-of-the-art drug testing pipeline that integrates PDOs with tumor-infiltrating lymphocytes (TILs). This platform accurately replicates real-time patient responses to chemotherapy, targeted treatments, and T-cell bispecific antibodies, highlighting the critical impact of the immune microenvironment on therapy efficacy (Kjølle et al, 2026, Cells)
Postoperative Therapeutic Windows
We investigate how physiological stress and lifestyle interventions impact cancer progression. Our preclinical studies reveal that a boost in physical activity specifically following surgery can double median survival time and significantly delay metastatic development, highlighting the postoperative period as a critical therapeutic window to counteract surgical stress and halt metastasis (Stagaard et al, 2025, Frontiers in Immunology).
III. Therapeutic Innovation & Drug Discovery
We translate ECM and microenvironment biology into new interventions designed to dismantle the metastatic niche. Current efforts include therapeutic approaches to target metastatic tumours, and development paths toward clinical translation and potential spin‑out.
Selected publications
- Kjølle S, Presti M, de Pina Roque J, Bisgaard LH, Ramos DB, Zornhagen KW, Yde CW, Schmidt AY, Verdys P, Højgaard M, Lassen U, Svane IM, Rohrberg KS, Donia M, Erler JT. Immune Cell Modulation of Patient-Matched Organoid Drug Response in Precision Cancer Medicine Platform. Cells. 2026 Jan 29;15(3):259. https://doi.org/10.3390/cells15030259
- Oria VO, Castro JL, de Pina Roque J, Martinez AG, Jørgensen FI, Lukassen MV, Rib L, Høg MKG, Mygind KJ, Erler JT. Crosstalk between tumor endothelial cells and cancer cells is important for metastasis initiation. Cell Commun Signal. 2025 Oct 17;23(1):443. doi: 10.1186/s12964-025-02441-3.https://doi.org/10.1186/s12964-025-02441-3
- Xia T, Zornhagen KW, Miinalainen I, Abramovitz L, Madsen CD, Nicolau M, Mayorca-Guiliani AE, Erez N, Erler JT. Loss of nidogen-1 causes lung basement membrane defects and increased metastasis. Front Immunol. 2025 Oct 16;16:1598547. doi: 10.3389/fimmu.2025.1598547.https://doi.org/10.3389/fimmu.2025.1598547
- Stagaard R, Jensen A, Schauer T, Bay ML, Tavanez ARA, Wielsøe S, Peletier M, Strøbech JE, Oria VO, Zornhagen KW, Albrechtsen R, Christensen JF, Erler JT. Exercise boost after surgery improves survival in model of metastatic breast cancer. Front Immunol. 2025 Feb 24;16:1533798.https://doi.org/10.3389/fimmu.2025.1533798
- Emerson MJ, Willacy O, Madsen CD, Reuten R, Brøchner CB, Lund TK, Dahl AB, Jensen THL, Erler JT, Mayorca-Guilliani AE. Machine learning identifies remodeling patterns in human lung extracellular matrix. Acta Biomater. 2025 Mar 15;195:94-103. https://doi.org/10.1016/j.actbio.2024.12.062
- Perryman L, Høye AM, Cox TR, Baker AM, Strøbech J, Leonte L, Singh LB, Popov S, Lorentzen LG, Reuten R, Poulsen HS, Davies MJ, Jones C, Erler JT. Cleavage of the vascular matrix attracts glioblastoma cells to infiltrate the brain parenchyma. BioRxiv. 2024. https://doi.org/10.1101/2024.08.02.606279
- Rafaeva M, Jensen ARD, Horton ER, Zornhagen KW, Strøbech JE, Fleischhauer L, Mayorca-Guiliani AE, Nielsen SR, Grønseth DS, Kuś F, Schoof EM, Arnes L, Koch M, Clausen-Schaumann H, Izzi V, Reuten R, Erler JT. Fibroblast-derived matrix models desmoplastic properties and forms a prognostic signature in cancer progression. Front Immunol. 2023 Jul 18;14:1154528. https://doi.org/10.3389/fimmu.2023.1154528
- Strøbech JE, Giuriatti P, Stagaard R, De Sepulveda P, Nielsen SR, Erler JT. FES null mice demonstrate a reduction in neutrophil dependent pancreatic cancer metastatic burden. Front Oncol. 2023 Mar 9;13:1096499.https://doi.org/10.3389/fonc.2023.1096499
- Rafaeva M, Horton ER, Jensen ARD, Madsen CD, Reuten R, Willacy O, Brøchner CB, Jensen TH, Zornhagen KW, Crespo M, Grønseth DS, Nielsen SR, Idorn M, Straten PT, Rohrberg K, Spanggaard I, Højgaard M, Lassen U, Erler JT, Mayorca-Guiliani AE. Modeling Metastatic Colonization in a Decellularized Organ Scaffold-Based Perfusion Bioreactor. Adv Healthc Mater. 2022 Jan;11(1):e2100684.https://doi.org/10.1002/adhm.202100684
- Reuten R, Zendehroud S, Nicolau M, Fleischhauer L, Laitala A, Kiderlen S, Nikodemus D, Wullkopf L, Nielsen SR, McNeilly S, Prein C, Rafaeva M, Schoof EM, Furtwängler B, Porse BT, Kim H, Won KJ, Sudhop S, Zornhagen KW, Suhr F, Maniati E, Pearce OMT, Koch M, Oddershede LB, Van Agtmael T, Madsen CD, Mayorca-Guiliani AE, Bloch W, Netz RR, Clausen-Schaumann H, Erler JT. Basement membrane stiffness determines metastases formation. Nat Mater. 2021 Jun;20(6):892-903.https://doi.org/10.1038/s41563-020-00894-0
- Nielsen SR, Strøbech JE, Horton ER, Jackstadt R, Laitala A, Bravo MC, Maltese G, Jensen ARD, Reuten R, Rafaeva M, Karim SA, Hwang CI, Arnes L, Tuveson DA, Sansom OJ, Morton JP, Erler JT. Suppression of tumor-associated neutrophils by lorlatinib attenuates pancreatic cancer growth and improves treatment with immune checkpoint blockade. Nat Commun. 2021 Jun 7;12(1):3414.https://doi.org/10.1038/s41467-021-23731-7
- Jensen ARD, Horton ER, Blicher LH, Pietras EJ, Steinhauer C, Reuten R, Schoof EM, Erler JT. Organ-Specific, Fibroblast-Derived Matrix as a Tool for Studying Breast Cancer Metastasis. Cancers (Basel). 2021 Jul 2;13(13):3331.https://doi.org/10.3390/cancers13133331
- Mayorca-Guiliani AE, Rafaeva M, Willacy O, Madsen CD, Reuten R, Erler JT. Decellularization of the Murine Cardiopulmonary Complex. J Vis Exp. 2021 May 30;(171).https://app.jove.com/v/61854/decellularization-of-the-murine-cardiopulmonary-complex
- Venning FA, Zornhagen KW, Wullkopf L, Sjölund J, Rodriguez-Cupello C, Kjellman P, Morsing M, Hajkarim MC, Won KJ, Erler JT, Madsen CD. Deciphering the temporal heterogeneity of cancer-associated fibroblast subpopulations in breast cancer. J Exp Clin Cancer Res. 2021 May 20;40(1):175.https://doi.org/10.1186/s13046-021-01944-4.
- Excellence with purpose — rigorous science aligned to patient impact.
- High‑performance teamwork — ambitious goals, clear ownership, supportive mentorship.
- Open innovation — collaboration across academia, hospitals, and industry to accelerate translation.
- Integrity and reproducibility — robust, transparent, data‑driven work.
- Growth mindset — continuous learning and leadership development at every career stage.
Janine’s leadership style is transformational. Backed by over 25 years of academic excellence, she cultivates psychological safety to build high trust, high performance teams, combining clear communication with dedicated mentorship.
As an innovation leader and experienced biotech Founder and CEO, she actively bridges the gap between rigorous scientific discovery and real world translation.
By fostering a culture of partnership and entrepreneurial growth, Janine is driven to turn bold ideas into tangible solutions that deliver measurable impact for patients, partners, and society.
- Danish Cancer Society
- Novo Nordisk Foundation
- Lundbeckfonden
- ERC - European Research Council
- Innovation Fund Denmark

We believe that great science requires great collaboration. Each year, our research group hosts a retreat at a summer house to foster scientific discourse and team cohesion.
This annual event provides a change of scenery to discuss ongoing projects, brainstorm future directions, and build lasting professional and personal connections through team-building activities.

The laboratory has been awarded the LEAF - Laboratory Efficiency Assessment Framework Silver certificate.


