Behrendt Group
Our major interest is the interplay between cells and their microenvironment in health and disease. We work to understand the interactions between cells and the extracellular matrix and to utilize this understanding to create novel therapeutic options, particularly in cancer treatment. Our particular focus is receptors and proteolytic enzymes that govern the homeostasis and degradation of collagen, the major protein constituent of higher organisms.
The Behrendt Group is located at the Finsen Laboratory, a cancer research department at the Finsen Centre at Rigshospitalet, Copenhagen University Hospital.
Our vision is to provide important contributions to the understanding of cancer invasion at the molecular level, to demonstrate novel molecular targets for cancer therapy with relevance all the way to the clinic and to perform such active steps in drug development that lead to the realization of the drug potential. To pursue this goal, we study the properties of molecular components engaged in extracellular matrix degradation and cancer invasion and utilize the knowledge obtained for development of novel means of cancer therapy, including targeted drug delivery.
- Molecular and functional characterization of the endocytic collagen receptor, uPARAP/Endo180
- Demonstration of the expression and pathological role of uPARAP/Endo180 in various cancer types and other diseases
- Demonstration of the functional interplay between endocytic collagen receptors and extracellular proteases in the breakdown of the extracellular matrix (ECM)
- Development of several antagonist antibodies against ECM degrading components, enabling model-based therapy studies in vivo
- Utilization of uPARAP/Endo180 as a novel cancer target to enable targeted drug delivery and development of a targeted model therapeutic with anti-cancer effect in vivo
Our basic research is focused on those proteins on the surface of cancer cells and cancer-associated non-malignant cells which play a direct role in cancer invasion. We have particular focus on receptors and proteolytic enzymes that take part in the breakdown of the extracellular matrix. The translational research includes the development of therapeutic targeting, using functional blocking of components active in invasion, and more recently by utilizing the same components for targeted drug delivery (Fig. 1).
Major project headings:
- Functional role of the endocytic receptors uPARAP/Endo180 and MR: Defining the ligand spectrum using mass spectrometry-based proteomics
- Understanding the role of uPARAP and extracellular proteases in osteosarcoma and other cancers
- Targeted drug delivery using uPARAP-directed antibody-drug conjugates (ADCs) (Fig 2), also including
- Novel disease indications (mesothelioma)
- Stromal targeting
- Novel ADC formats
Figure 1. Project themes
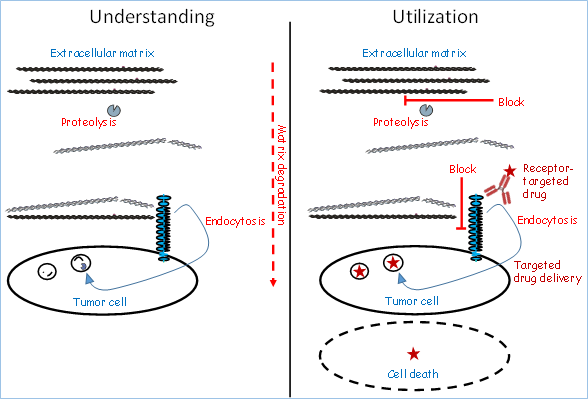
Our research program includes projects focused on the understanding of cancer invasion and matrix degradation, as well as translational projects aiming at the utilization of the knowledge obtained.
Left: The Understanding part includes studies on invasion-associated degradation processes in the ECM. Particular focus on proteases and endocytic receptors engaged in collagen turnover.
Right: The Utilization part is focused on a) the functional blocking of degradation reactions with importance for invasion; b) the exploitation of the studied endocytic receptors for targeted drug delivery, using a Trojan Horse strategy with ADCs.
Figure 2. Antibody-drug conjugates
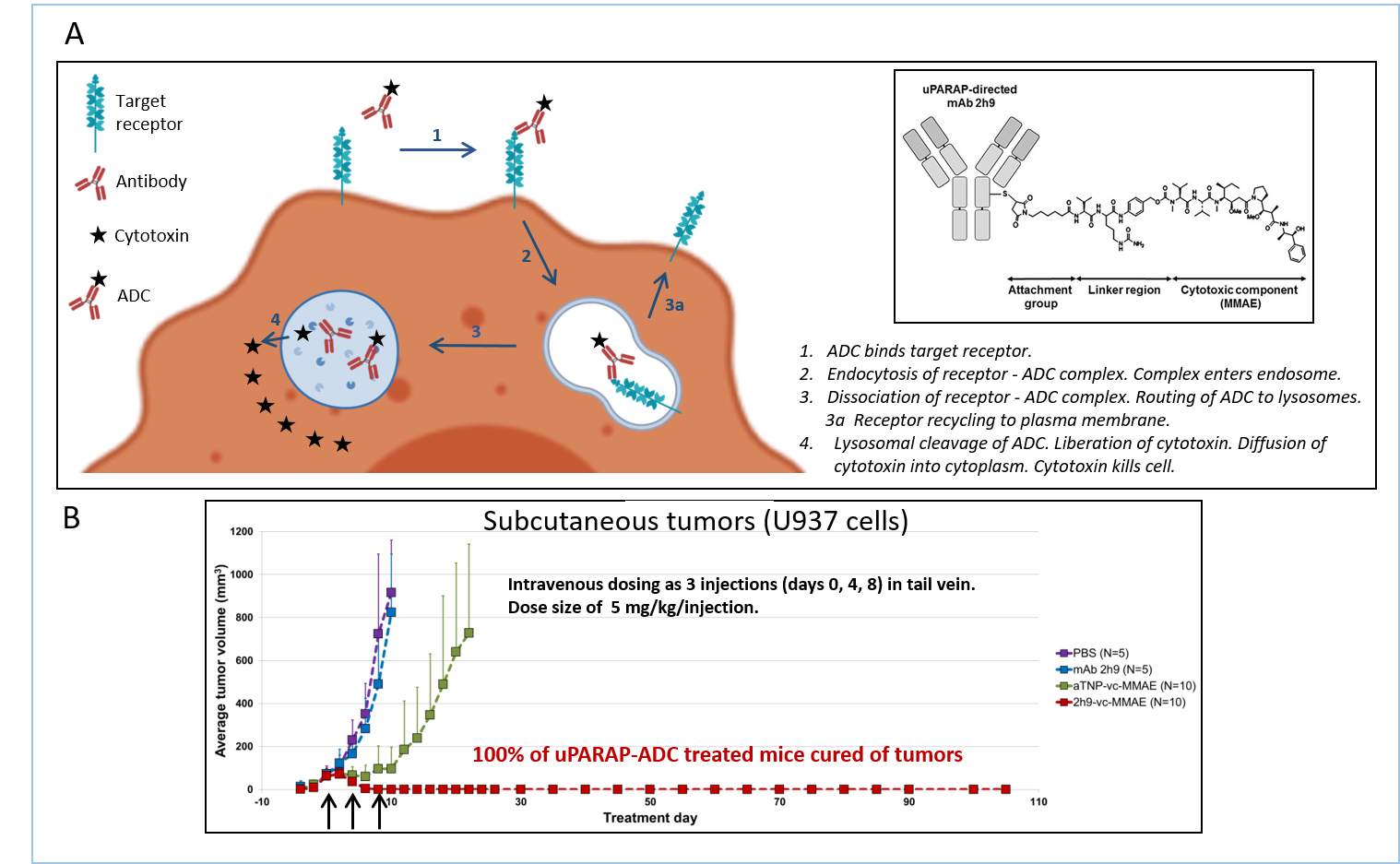
Antibody-drug conjugates (ADCs) are a rapidly emerging type of drugs. Several ADCs against various cancer targets have now been approved for use in the clinic while a much larger number are in preclinical and clinical development.
A Mode of action of ADCs and structure of a uPARAP-directed ADC. The receptor-specific antibody and the cytotoxin are connected through a covalent linker structure that is stable in the circulation but susceptible to cleavage in the lysosome.
B Treatment of mice carrying subcutaneous U937 tumors with uPARAP-directed ADC (2H9-vc-MMAE). From ref. Nielsen et al., 2017.
Key technologies include receptor-ligand studies (proteomics, flow cytometry, analyses of endocytosis), CRISPR/Cas-9-based gene manipulation, antibody and ADC technology, mouse tumor models and general technology for protein handling, cell-based assays and molecular biology.
Selected publications
Ingvarsen, S.Z., Gårdsvoll, H., van Putten, S., Nørregaard, K.S., Krigslund, O., Meilstrup, J.A., Tran, C., Jürgensen, H.J., Melander, M.C., Nielsen, C.H., Kjaer, A., Bugge, T.H., Engelholm, L.H. and Behrendt, N. Tumor cell MT1-MMP is dispensable for osteosarcoma tumor growth, bone degradation and lung metastasis. Sci. Rep. (2020), 10:19138.
Jürgensen, H.J., Nørregaard, K.S., Sibree, M.M., Santoni-Rugiu, E., Madsen, D.H., Wassilew, K., Krustrup, D., Garred, P., Bugge, T.H., Engelholm, L.H. and Behrendt, N. Immune regulation by fibroblasts in tissue injury depends on uPARAP-mediated uptake of collectins. J. Cell Biol. (2019), 218: 333-349.
Nielsen, C.F., van Putten, S.M., Lund, I.K., Melander, M.C., Nørregaard, K.S., Jürgensen, H.J., Reckzeh K., Christensen, K.R., Ingvarsen, S.Z., Gårdsvoll, H., Jensen, K.E., Hamerlik, P., Engelholm, L.H. and Behrendt, N. The collagen receptor uPARAP/Endo180 as a novel target for antibody-drug conjugate mediated treatment of mesenchymal and leukemic cancers. Oncotarget (2017), 8: 44605-44624.
Engelholm, L.H., Melander, M.C., Hald, A., Persson, M., Madsen, D.H., Jürgensen, H.J., Johansson, K., Nielsen, C., Nørregaard, K.S., Ingvarsen, S.Z., Kjær, A., Trovik, C.S., Lærum, O.D., Bugge, T.H., Eide, J. and Behrendt, N. Targeting a novel bone degradation pathway in primary bone cancer by inactivation of the collagen receptor uPARAP/Endo180. J. Pathol. (2016), 238: 120-133.
Ingvarsen, S., Porse, A., Erpicum, C., Maertens, L., Jürgensen, H.J., Madsen, D.H., Melander, M.C., Gårdsvoll, H., Høyer-Hansen, G., Noel, A., Holmbeck, K., Engelholm, L.H. and Behrendt, N. Targeting a single function of the multifunctional matrix metalloprotease MT1-MMP: impact on lymphangiogenesis. J. Biol. Chem. (2013), 288: 10195-10204.
In the media
Denmark funds BRIC cancer research projects
17 November 2023, on bric.ku.dk
Tumor targeting
Our results with tumor targeting have been treated in articles in the Newsletter of the Danish Cancer Society (2015), “Dagens Medicin” (2015) and “Medwatch” (2016). The spin‐out process, moving the ADCendo company into the Creation House program at the BioInnovation Institute, has been taken up in the Newsletter at Sund, University of Copenhagen.
Most central current collaborations:
Group leader Lars H. Engelholm (Finsen Laboratory / BRIC, Univ. Cph., DK): Translational research projects including targeted drug delivery. Senior investigator Thomas H. Bugge (NIH, USA): ECM degradation and cancer invasion. Project manager Christoffer F. Nielsen (Adcendo ApS, DK): ADC technology. Professor Agnès Noel (Univ. Liege, BE): Lymphangiogenesis. Consultant Eric Santoni‐Rugiu (Rigshospitalet / BRIC, Univ. Cph., DK): Lung pathology and mesothelioma. Junior group leader Daniel H. Madsen (CCIT, Herlev Hospital, DK): Cell‐matrix interactions and immunological aspects. Professor Fredrik Björkling (Dept. Drug Design & Pharmacology, Univ. Cph., DK) and Assistant professor Katrine Qvortrup (Dept. Chemistry, Technical University DK): ADC cytotoxins and linkers. Participation in the DCCC Brain Tumor Center (DK).








